Module 19: Spitz and Reed Nevi
1. Learning Objectives
After completing this module, the learner should be able to:
- Distinguish between Spitz nevus and Reed nevus based on dermoscopic morphology, pigmentation, and cellular composition while recognizing that they exist along a shared spectrum.
- Identify the three main dermoscopic patterns of Spitz/Reed nevi --- starburst, globular with negative network, and dotted-vessel patterns --- and describe which lesion subtype each pattern exemplifies.
- Recognize the dermoscopic features of nonpigmented (classic) Spitz nevus, including dotted vessels, negative network, and shiny white lines (white network/reticular depigmentation).
- Describe the natural dermoscopic evolution of Spitz/Reed nevi from globular pattern through starburst to homogeneous/reticular senescence, including the possibility of spontaneous involution.
- Identify dermoscopic features that distinguish spitzoid melanoma from benign Spitz/Reed nevi, including asymmetric distribution of structures and colors, multicomponent patterns, and the critical significance of patient age.
- Apply the three-step management algorithm for spitzoid-looking lesions incorporating dermoscopic symmetry assessment, patient age (12-year threshold), and clinical morphology (flat vs. nodular).
- Describe the concept, classification theories, and dermoscopic characteristics of atypical Spitz tumors (ASTs) and explain why dermoscopy alone cannot reliably distinguish ASTs from classic Spitz nevi.
- Formulate an appropriate monitoring protocol for spitzoid-looking lesions in children, including follow-up intervals, criteria for excision, and criteria for monitoring termination.
2. Prerequisites
| Module | Title | Relevance |
|---|---|---|
| Module 01 | Introduction and Principles of Dermoscopy | Polarized vs. nonpolarized dermoscopy; shiny white lines are visible only with polarized light |
| Module 02 | Histopathologic Correlations of Dermoscopic Structures | Understanding of streaks, pseudopods, globules, dots, negative network, blue-whitish veil, and their histopathologic correlates |
Recommended additional background:
| Module | Title | Relevance |
|---|---|---|
| Module 03 | Pattern Analysis Revised | Fundamental pattern recognition concepts |
| Module 04 | Top-Down 2-Step Algorithm | Organized vs. disorganized pattern assessment |
| Module 16 | Acquired Melanocytic Nevi | Baseline understanding of common globular and reticular nevi for comparison |
3. Key Concepts
3.1 Historical Context
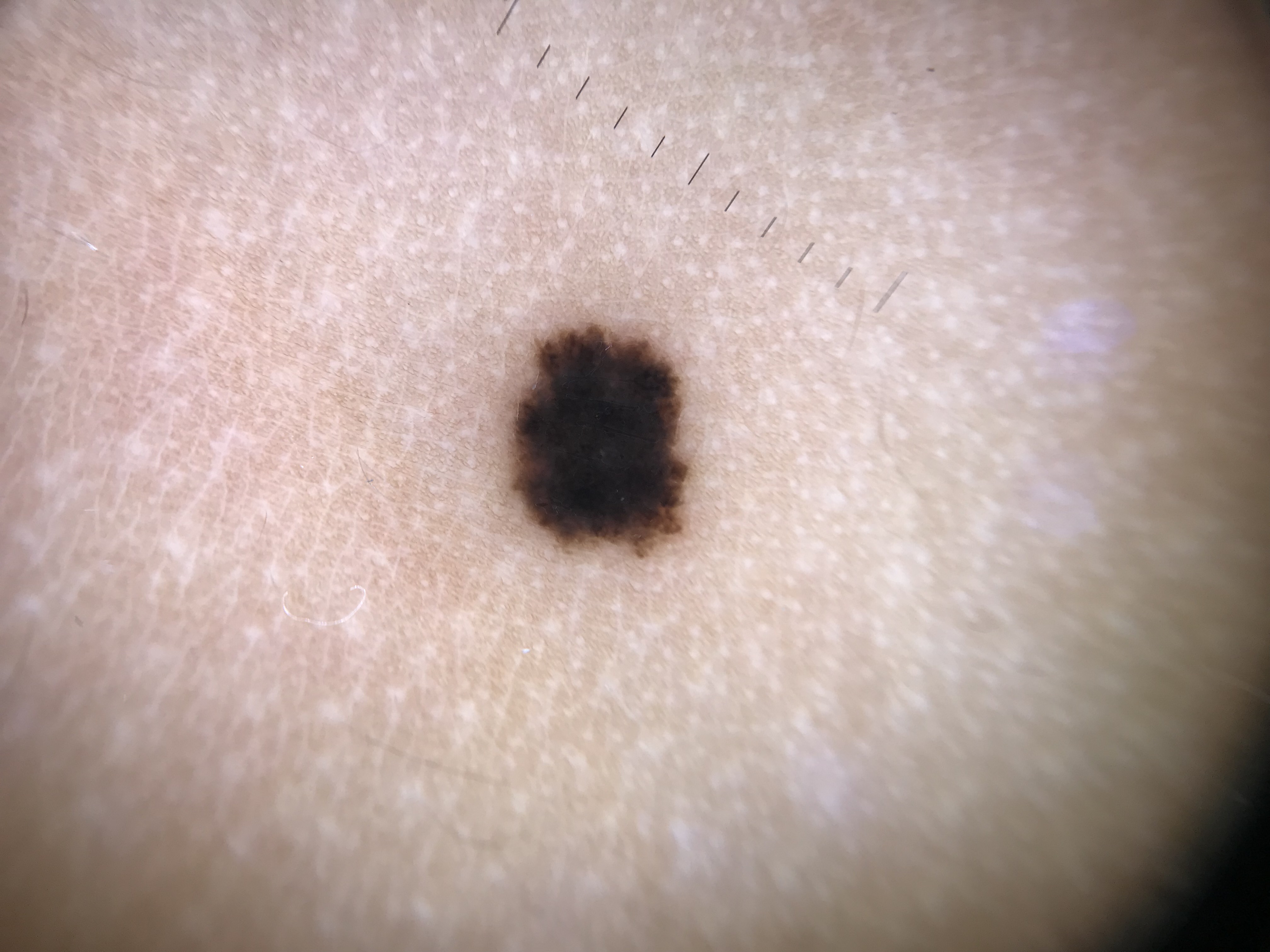
- Sophie Spitz (1948): Described a distinct type of melanocytic tumor in childhood displaying epithelioid and spindle cells, originally termed "melanoma of childhood." The lesion now bears her name --- Spitz nevus.
- Richard Reed: Later described a pigmented variant --- the pigmented spindle cell nevus (spindle cell and/or epithelioid cell nevus), now known as Reed nevus.
3.2 Spitz Nevus vs. Reed Nevus

| Feature | Spitz Nevus | Reed Nevus |
|---|---|---|
| Pigmentation | Can be nonpigmented (amelanotic) or pigmented | Heavily pigmented (dark brown to black) |
| Cellularity | Epithelioid and spindle cells | Predominantly spindle cells |
| Classic dermoscopic pattern | Dotted vessels + white network (nonpigmented); globular + negative network (pigmented) | Starburst pattern (symmetric peripheral streaks/pseudopods) |
| Relationship | Originally described as separate entity | Originally described as a pigmented variant of Spitz nevus |
3.3 The Spitzoid Spectrum
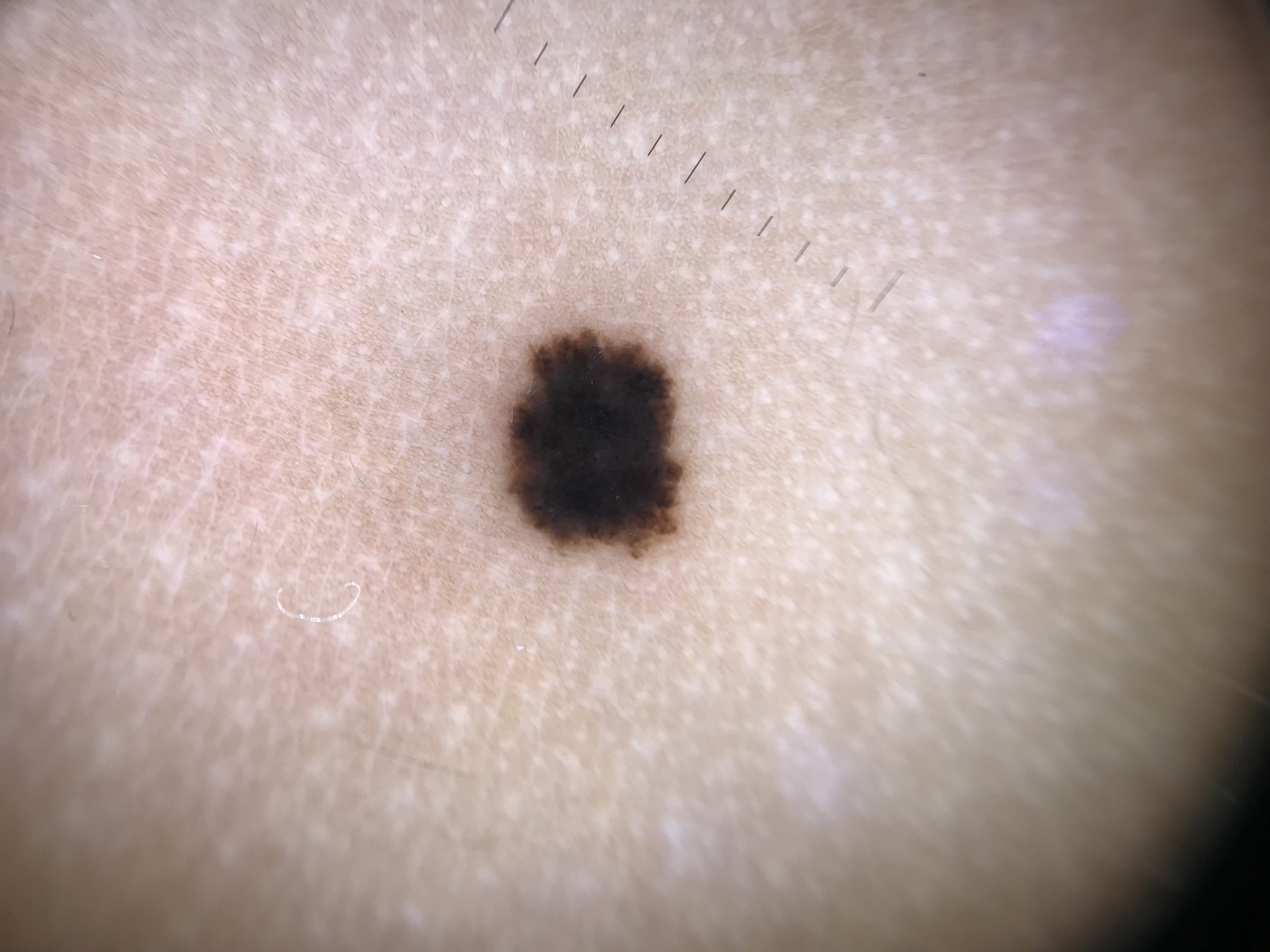
Although Reed nevus was historically considered a pigmented variant of Spitz nevus, many researchers now regard Spitz and Reed nevi as distinct entities based on differences in age predilection, anatomic site preference, and other features. Nonetheless, based on cellular morphology, they appear to belong to the same spectrum.
The core problem: The biological behavior, natural evolution, and malignant potential of Spitz/Reed nevi have not been fully elucidated. Melanomas can resemble Spitz/Reed nevi clinically, dermoscopically, and even histopathologically. No single test or combination of tests can reliably differentiate Spitz/Reed nevi from melanoma in all cases.
3.4 Terminology for Ambiguous Spitzoid Lesions
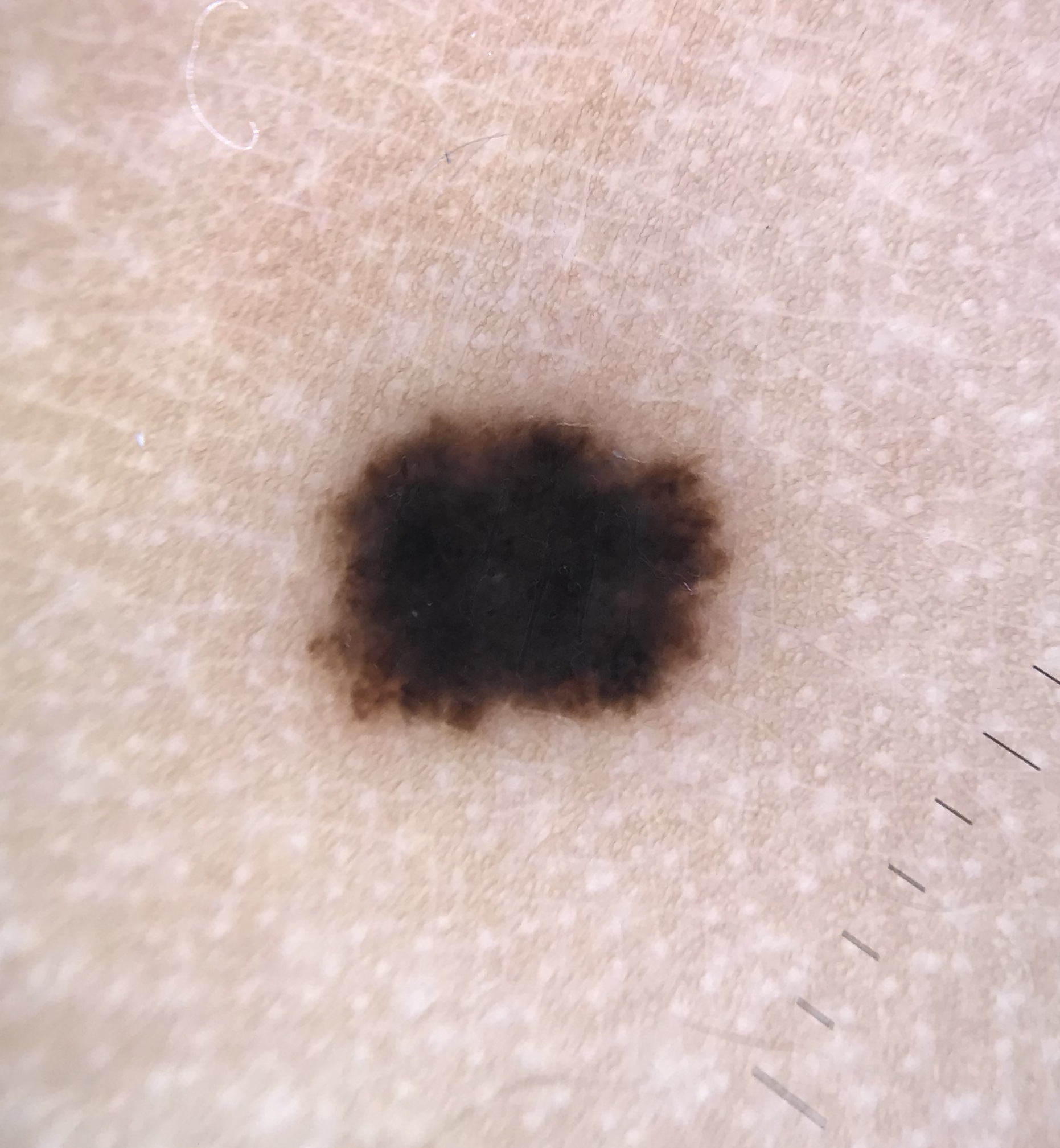
The following terms are used for spitzoid tumors that cannot be confidently classified as benign or malignant:
- Atypical Spitz tumors (ASTs)
- Atypical Spitz nevus
- Spitz nevus with atypia and metastasis
- Metastasizing Spitz tumor
- Melanocytic tumors of unknown malignant potential (MELTUMP)
- Melanocytoma
4. Core Content
4.1 Dermoscopy of Spitz/Reed Nevi --- Overview
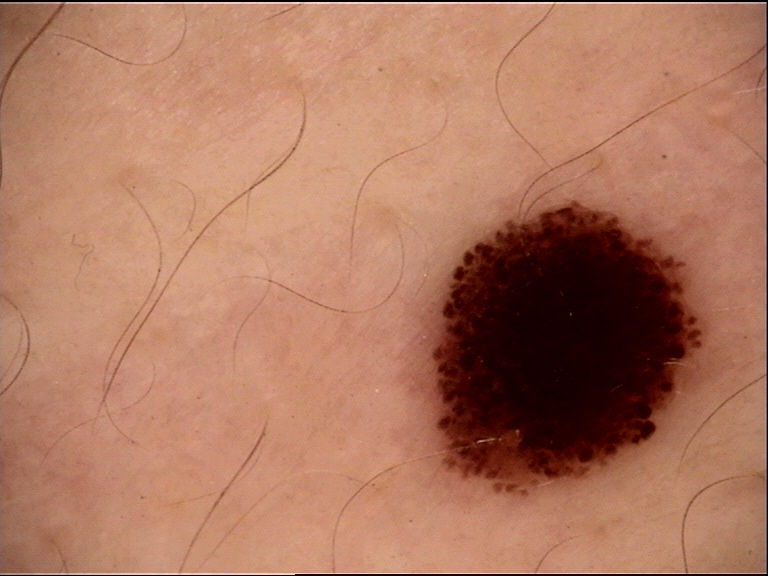
Dermoscopy of Spitz and Reed nevi reveals a few unique and reproducible findings that enable their clinical recognition --- many of which would not have been diagnosed otherwise. While existing controversies regarding the biologic nature of spitzoid tumors have not been solved, dermoscopy has provided insights that have influenced management.
A systematic review by the International Dermoscopy Society (Lallas et al., 2017) analyzed 896 Spitz nevi from 15 studies and found the following pattern distribution:
| Dermoscopic Pattern | Frequency |
|---|---|
| Starburst | 51% |
| Dotted vessels | 19% |
| Globular | 17% |
| Multicomponent/atypical | 9% |
| Other patterns | 4% |
Age-related variation (de Mestier et al., 2017): In younger patients (12 years and under), Spitz nevi are more likely to exhibit a globular and nonpigmented homogeneous pattern. Starburst and reticular patterns are more commonly observed on the anterior trunk and acral sites.
4.2 Classic Nonpigmented Spitz Nevus

4.2.1 Typical Dermoscopic Pattern: Dotted Vessels + White Network
"Classic" nonpigmented Spitz nevi are dermoscopically typified by a pattern of multiple, regularly distributed, dotted vessels. The hallmarks include:
- Dotted vessels: Evenly distributed in a symmetric arrangement. This is the defining vascular pattern.
- Globular/glomerular vessels: In more raised or nodular lesions, vessels tend to be larger and may appear as globular or glomerular vessels.
- Additional vessel types: Comma, hairpin, serpentine, and corkscrew vessels can also be seen in nodular Spitz nevi.
- Negative network (inverse network): Elongated, curvilinear globules that, when aggregated, create a negative (inverse) pigment network. Visible in both pigmented and some nonpigmented variants.
- Shiny white lines (polarized light only): Crisscrossing white lines forming a white network (also termed reticular depigmentation). In nonpigmented Spitz nevi, the shiny white lines of the network appear to surround the vessels.
Key diagnostic combination: The co-occurrence of regularly distributed dotted vessels combined with shiny white lines (white network) in a symmetric arrangement is the exemplar pattern of classic nonpigmented Spitz nevus.
4.2.2 Symmetry as a Defining Feature
The typical dermoscopic pattern of nonpigmented Spitz nevi requires symmetric distribution of both vascular structures and shiny white lines. This symmetry is critical for distinguishing benign from potentially malignant lesions.
4.2.3 Atypical Nonpigmented Spitz Nevi
Nonpigmented Spitz nevi that lack symmetric distribution of structures also exist, including:
- Angiomatous Spitz nevi: Prominent vascular component
- Desmoplastic Spitz nevi: Fibrotic/sclerotic features
Critical warning: A Spitz nevus lacking dermoscopic symmetry cannot be differentiated from melanoma. Any spitzoid-looking lesion that displays an asymmetric distribution of structures should be biopsied and examined histopathologically.
Check Your Understanding
What is the classic dermoscopic pattern of a Spitz nevus?
The classic Spitz nevus shows a starburst pattern with symmetrically distributed streaks (pseudopods and radial lines) at the periphery, often surrounding a central hypopigmented or pink structureless area. The symmetry of the peripheral projections is the hallmark that distinguishes benign Spitz from melanoma.
Key Takeaways
- The starburst pattern (symmetrical peripheral streaks/pseudopods radiating in all directions) is the hallmark of classic Spitz/Reed nevi, indicating active peripheral junctional growth.
- Reed nevus (pigmented spindle cell nevus) typically shows a starburst pattern with dark brown-to-black color, while classic Spitz is often pink-red with subtle features.
- The globular pattern of Spitz nevus (symmetric peripheral globules) is common in children and represents the growth phase of a benign Spitz proliferation.

Clinical Scenario
A 7-year-old girl presents with a 5 mm pink, dome-shaped papule on her left cheek that appeared 4 months ago and has been slowly growing. Dermoscopy reveals multiple regularly distributed dotted vessels in a symmetric arrangement with shiny white lines forming a white network pattern under polarized light.
What is the diagnosis, and how does patient age influence management?
Classic Nonpigmented Spitz Nevus
The symmetric dotted vessels with shiny white lines (white network) is the exemplar pattern of a classic nonpigmented Spitz nevus. In a child under 12, this symmetric pattern strongly favors a benign Spitz nevus. Management per the three-step algorithm: the lesion is symmetric, the patient is under 12, and the lesion is nodular but small -- dermoscopic monitoring at 3-month intervals is appropriate. If the same lesion presented in an adult, excision would be recommended regardless of symmetry due to the increased risk of spitzoid melanoma with age.
4.3 Pigmented Spitz Nevus

4.3.1 Globular Pattern with Negative Network
Pigmented Spitz nevi are dermoscopically typified by the presence of globules:
- Varied globules: Globules that vary in size and color, ranging from different shades of brown to gray and black. This variability is a key distinguishing feature.
- Elongated/curvilinear globules: Can appear elongated and curvilinear, and when aggregated, create the negative network.
- Negative network: Formed by the aggregation of irregular globules, creating a "negative" or "inverse" network pattern.
- Shiny white lines: May also be present. The co-existence of negative network and shiny white lines is very helpful for recognition and is not seen in classic dermal globular nevi.
4.3.2 Differentiation from Classic Dermal Nevi
| Feature | Pigmented Spitz Nevus | Classic Dermal Globular Nevus |
|---|---|---|
| Globule size | Variable (varying sizes) | Uniform |
| Globule color | Multiple shades (brown, gray, black) | Monomorphous brown |
| Negative network | Present | Absent |
| Shiny white lines | Often present | Absent |
4.3.3 Starburst Pattern in Pigmented Spitz Nevi
Some researchers suggest that pigmented Spitz nevi are typified by a globular pattern while Reed nevi are typified by a starburst pattern. However, this distinction is not absolute. The starburst pattern in Spitz nevi can manifest with:
- Peripheral tiered globules: Globules layered one on top of another at the periphery
- Peripheral streaks: Streaks arranged around the entire perimeter
Note: Although the starburst pattern composed of peripheral tiered globules is commonly seen in Spitz nevi, it can also be seen in atypical/dysplastic nevi with spitzoid features.
4.4 Reed Nevus (Pigmented Spindle Cell Nevus)
4.4.1 The Starburst Pattern --- Classic Feature
Reed nevus is dermoscopically typified by the starburst pattern, named for its resemblance to an exploding star. Its components are:
- Central area: Dark brown/black/blue pigmentation, often homogeneous
- Peripheral streaks: Multiple pigmented streaks (including both radial streaming and pseudopods) that arise from the center and radially expand toward the periphery of the lesion
- Mandatory symmetry: A starburst pattern always presupposes symmetry in terms of distribution of colors and structures
Histopathologic correlation: The streaks/pseudopods correspond to confluent, elongated nests of melanocytes located at the dermoepidermal junction.
4.4.2 Additional Dermoscopic Patterns in Reed Nevi
Beyond the classic starburst, Reed nevi may display:
| Pattern | Description |
|---|---|
| Homogeneous | Structureless pattern of diffuse brown, gray-blue to gray-black pigmentation in the absence of other discernible structures |
| Reticular (superficial black network) | Prominent dark pigment network with an underlying bluish haze covering most parts of the lesion |
4.4.3 Reed Nevus Evolution
Longitudinal follow-up of Reed nevi has demonstrated:
- Starburst pattern corresponds to the radial growth phase (active growth)
- Senescence: When a Reed nevus enters senescence, it usually manifests a homogeneous pattern but can also display a reticular pattern
- Recognition challenge: At the senescent stage, recognition is difficult because several other types of nevi may exhibit similar homogeneous or reticular patterns
Check Your Understanding
What dermoscopic features differentiate a Reed nevus from a Spitz nevus?
Reed nevus (pigmented spindle cell nevus) shows a symmetric starburst pattern with prominent blue-black to dark brown streaks radiating from the periphery, often with a dark central blotch. Unlike Spitz nevus, Reed nevi are deeply pigmented. The key shared feature is symmetry, but Reed nevi are characteristically darkly pigmented while Spitz nevi are often pink or lightly pigmented.
4.5 Summary of the Three Main Dermoscopic Patterns
| Pattern | Exemplar Lesion | Key Features |
|---|---|---|
| 1. Starburst | Reed nevus | Central dark pigmentation + symmetric peripheral streaks/pseudopods |
| 2. Globular + negative network + shiny white lines | Pigmented Spitz nevus | Variable-sized globules, reticular depigmentation, shiny white lines |
| 3. Dotted vessels + white network | Nonpigmented (classic) Spitz nevus | Regularly distributed dotted vessels surrounded by shiny white lines |
Key Takeaways
- Atypical spitzoid tumors show asymmetric structure, irregular streaks/pseudopods, blue-white veil, or atypical vascular patterns that overlap with melanoma.
- Negative network (serpiginous hypopigmented lines surrounding globules) is seen in both Spitz nevi and melanoma; distribution symmetry is the key differentiating factor.
- Age is a critical modifier: starburst or globular pattern in children strongly favors Spitz nevus, while the same features in adults over 40 raise melanoma concern.

Clinical Scenario
A 25-year-old woman presents with a 4 mm dark brown-black papule on her right thigh that appeared 6 months ago. Dermoscopy reveals a striking starburst pattern with symmetric peripheral streaks radiating in all directions from a homogeneous dark brown-black center.
What is the most likely diagnosis, and how does management differ from pediatric cases?
Reed Nevus (Pigmented Spindle Cell Nevus)
The symmetric starburst pattern with dark brown-black pigmentation is the classic dermoscopic appearance of a Reed nevus. However, in a 25-year-old adult, management differs from a child: the three-step algorithm recommends excision for any spitzoid-looking lesion in patients over 12, even when symmetric, because melanoma can present with a starburst pattern in adults. A starburst pattern in a child under 12 would be monitored; in an adult, excision is strongly recommended to obtain histopathologic confirmation.
4.6 Multicomponent Pattern
Some Spitz/Reed nevi present with a multicomponent pattern, composed of more than one of the three main patterns described above.
Critical management implication: A pigmented lesion displaying a multicomponent pattern should always be considered suspicious for melanoma, and biopsy should be performed on all such lesions.
Check Your Understanding
When should a Spitz/Reed nevus be excised versus monitored?
Excision is recommended for: asymmetric Spitz nevi (atypical spitzoid lesion), any spitzoid lesion in adults (where de novo Spitz nevi are uncommon), nodular growth, ulceration, or size >1 cm. Monitoring may be appropriate for classic symmetric Spitz nevi in children, where the starburst pattern is expected to evolve into a homogeneous or globular pattern over time.
4.7 Evolution of Spitz Nevi
Sequential digital monitoring has revealed a predictable evolutionary sequence for Spitz/Reed nevi:
Stage 1: Early Nevogenesis
Dense GLOBULAR pattern
|
v
Stage 2: Active Growth
Transformation to STARBURST pattern
(considered the exemplar, easy-to-identify stage)
|
v
Stage 3: Senescence
HOMOGENEOUS or RETICULAR pattern
(central blue-white veil may appear)
|
v
Stage 4: Terminal Phase
Retain reticular/homogeneous pattern
OR
Undergo spontaneous INVOLUTION
(partial or complete, over months to years)
Key insight: The starburst pattern, while considered the exemplar dermoscopic pattern of Spitz/Reed nevi, probably simply reflects an easy-to-identify stage in the natural evolution of these nevi. It has been suggested that the globular pattern might represent the initial stage of evolution of a pigmented Spitz nevus.
4.8 Dermoscopy of Spitzoid Melanoma
"Spitzoid" is a term adopted to describe melanomas with similar clinical and dermoscopic characteristics to those associated with Spitz nevi.
4.8.1 Shared Features with Spitz Nevi
Spitzoid melanomas often exhibit features also seen in Spitz nevi:
- Streaks
- Globules
- Dotted vessels
- Negative network
- Shiny white lines
4.8.2 Distinguishing Features of Spitzoid Melanoma
| Feature | Spitzoid Melanoma | Spitz/Reed Nevus |
|---|---|---|
| Color distribution | Multiple colors, disorganized | More uniform |
| Structural distribution | Disorganized/asymmetric | Symmetric/organized |
| Overall pattern | Multicomponent, asymmetric | Typically symmetric |
4.8.3 Exceptions and Overlap (Critical)
- Approximately 13% of dermoscopically symmetric spitzoid lesions in patients over 12 years of age are in fact melanomas.
- Some melanomas perfectly mimic symmetric Spitz/Reed nevi, rendering differentiation impossible.
- Some Spitz nevi display an atypical/multicomponent pattern most often associated with melanoma.
- Dermoscopic atypia of Spitz nevi does not necessarily correspond with histologic atypia. A Spitz nevus that looks alarming on dermoscopy may be histologically benign, and vice versa.
Key Takeaways
- Spitz nevi undergo predictable involution: starburst transforms to globular, then homogeneous, and finally reticular or structureless over months to years.
- Short-term digital dermoscopy (3-month follow-up) can be used for equivocal spitzoid lesions in children; symmetric growth supports a benign diagnosis.
- Any spitzoid lesion in an adult over 12 years that shows asymmetry, ulceration, or progressive focal change should be excised and submitted for expert histopathologic review.
4.9 Atypical Spitz Tumors (ASTs)
4.9.1 Definition
ASTs are melanocytic proliferations with intermediate histopathologic features between Spitz nevi and spitzoid melanoma, and their malignant potential is uncertain.
4.9.2 Three Prevailing Theories
| Theory | Description |
|---|---|
| Binary theory | ASTs are in fact either benign or malignant; the inability to classify is a matter of human diagnostic uncertainty |
| Spectrum theory | A continuous biologic spectrum exists from benign to malignant, with intermediate-potential lesions in between. Envisioned classification: typical Spitz nevus (benign) --> atypical Spitz nevus (benign, atypical histology) --> AST (potential low-grade melanoma) --> spitzoid melanoma (malignant) |
| Heterogeneous group theory | ASTs are a heterogeneous group of melanocytic tumors, some of low and others of high risk for malignant degeneration |
4.9.3 Dermoscopy of ASTs
A multicentric study (Moscarella et al., 2015) of 55 ASTs and 110 Spitz nevi found:
| AST Dermoscopic Pattern | Frequency | Clinical Implication |
|---|---|---|
| Multicomponent | 44% | Generally suggestive of melanoma |
| Nonspecific | 20% | Generally suggestive of melanoma |
| Nonpigmented typical spitzoid (dotted vessels +/- reticular depigmentation) | 20% | Mimics classic Spitz nevus |
| Other patterns | 16% | Variable |
Critical finding: 20% of ASTs mimic classic Spitz nevi by displaying a nonpigmented typical spitzoid pattern (regularly distributed dotted vessels with or without reticular depigmentation). Dermoscopy appears insufficient to discriminate between Spitz nevi and ASTs when both display this typical spitzoid pattern.
4.10 Age-Dependent Considerations
| Factor | Children (under 12 years) | Adolescents and Adults (12+ years) |
|---|---|---|
| Spitz nevus incidence | Most common age group for development | Extremely rare after 4th decade |
| Melanoma incidence | Exceedingly rare before puberty | Incidence increases with age |
| Pre-test probability | Spitzoid lesion much more likely benign | Significant melanoma risk; ~13% of symmetric spitzoid lesions are melanoma |
| Management approach | Monitoring may be appropriate for symmetric, flat lesions | Excision recommended or close monitoring |
| Pattern predominance | More likely globular and nonpigmented homogeneous | More varied patterns |
4.11 Management Algorithm for Spitzoid-Looking Lesions
Management decisions aim to eliminate the risk of missing a melanoma while minimizing the removal of benign lesions. The three-step algorithm addresses three key factors:
flowchart TD
A[Spitzoid-Looking<br/>Lesion Identified] --> B{Step 1: Dermoscopic<br/>Symmetry?}
B -->|Asymmetric /<br/>Disorganized| C[EXCISE to Rule<br/>Out Melanoma]
B -->|Symmetric /<br/>Organized| D{Step 2: Patient<br/>Age?}
D -->|Age >= 12 years| E[EXCISE or<br/>Close Monitoring]
D -->|Age < 12 years| F{Step 3: Clinical<br/>Morphology?}
F -->|Nodular| G[EXCISE or Close<br/>Monitoring Every 15 Days]
F -->|Flat / Macular| H[MONITOR Every<br/>3 Months]
G -->|Asymmetric growth| C
G -->|Remains symmetric| I[Prolong Intervals:<br/>1 mo, then 2 mo]
H -->|Stable 6 months| J[Terminate<br/>Monitoring]
H -->|Asymmetric change| C
I -->|Stabilized or<br/>involuted| J
Step 1: Assessment of Dermoscopic Symmetry
Text version of algorithm
Is the lesion dermoscopically SYMMETRIC/ORGANIZED?
| |
YES NO
| |
v v
Proceed to EXCISE to rule
Step 2 out melanoma
Rationale: Any dermoscopically asymmetric or disorganized lesion with spitzoid features (streaks, dotted vessels, or negative network) should be excised.
Step 2: Consideration of Patient Age
Is the patient YOUNGER than 12 years?
| |
YES NO (12 years or older)
| |
v v
Proceed to EXCISE or
Step 3 closely MONITOR
Rationale: A considerable proportion of symmetric spitzoid-appearing lesions developing after age 12 prove to be melanomas.
Step 3: Evaluation of Clinical Morphology (Flat vs. Nodular)
Is the lesion NODULAR or FLAT?
NODULAR (symmetric, in child <12): FLAT (symmetric, in child <12):
| |
v v
EXCISE or CLOSE MONITORING MONITOR until stabilization
- Every 15 days initially - Every 3 months initially
- Excise if asymmetric growth - Can prolong if remains symmetric
- If remains symmetric, prolong - Terminate after stabilization
to 1 month, then 2 months (no change for 6 months)
- Terminate when stabilized - Alternative: some experts argue
(6 months no change) or to simply IGNORE flat, pigmented
involuted symmetric Spitz/Reed in
prepubertal patients
- Another viewpoint: excise while
small to avoid larger scar later
Monitoring Termination Criteria
- Stabilization: No dermoscopically detectable changes between two sequential visits separated by at least 6 months
- Involution: Complete disappearance of the lesion
Limitations of Monitoring
- Approximately 20% of symmetric Spitz/Reed nevi in children will ultimately be excised because of asymmetric changes noted during follow-up.
- None of these excised lesions in the referenced study proved to be AST or melanoma.
- There is currently no way to predict which Spitz/Reed nevi will grow significantly larger or grow in an asymmetric manner.
- If a Spitz/Reed nevus is detected while small enough for simple excision, some argue it would be best to remove it at that time, because the resulting small scar is preferable to waiting.
4.12 Differential Diagnosis
4.12.1 Spitzoid Melanoma vs. Spitz/Reed Nevus
| Feature | Favors Spitz/Reed Nevus | Favors Spitzoid Melanoma |
|---|---|---|
| Symmetry | Symmetric/organized pattern | Asymmetric/disorganized pattern |
| Colors | Uniform or limited palette | Multiple colors |
| Structure distribution | Regular, organized | Irregular, haphazard |
| Age | Children (<12 years) | Adolescents and adults (>12 years) |
| Growth | Symmetric growth (may be rapid) | Asymmetric growth |
| Dermoscopic atypia | Can be present (does NOT equal histologic atypia) | Usually present |
| Pattern | Classic starburst, globular, or dotted vessel | Multicomponent, atypical |
Important caveat: These are trends, not absolute criteria. Exceptions exist in both directions. Dermoscopy and clinical morphology are NOT sufficient to reliably differentiate Spitz/Reed nevi from spitzoid melanoma in all cases.
4.12.2 Broader Differential Diagnosis for Spitzoid Lesions
| Diagnosis | Differentiating Dermoscopic Clues |
|---|---|
| Classic dermal/globular nevus | Monomorphous brown globules (uniform size/color); lacks negative network and shiny white lines |
| Atypical/dysplastic nevus with spitzoid features | May show peripheral tiered globules (starburst); evaluate in clinical context |
| Nodular melanoma | Atypical vessels, ulceration, blue-black homogeneous pattern; asymmetric structures |
| Pyogenic granuloma | Reddish homogeneous area with white collarette; rapid growth |
| Amelanotic melanoma | Polymorphous vessels; milky-red areas; shiny white structures |
Key Takeaways
- Complete excision is recommended for atypical spitzoid tumors at any age because histologic distinction from spitzoid melanoma can be extremely difficult.
- In children, symmetric spitzoid lesions can be monitored with serial dermoscopy, but parental counseling about the monitoring protocol is essential.
- BAP1-inactivated melanocytic tumors may present with spitzoid morphology and require immunohistochemistry (BAP1 staining) for definitive classification.
5. Spitz/Reed Pattern Reference Table
| Lesion Subtype | Primary Pattern | Key Structures | Symmetry | Pigmentation | Vessel Pattern |
|---|---|---|---|---|---|
| Nonpigmented Spitz (classic) | Dotted vessels + white network | Dotted vessels, shiny white lines, negative network | Symmetric | Amelanotic/pink | Dotted (may be globular/glomerular if nodular; comma, hairpin, serpentine, corkscrew in nodular variants) |
| Pigmented Spitz | Globular + negative network | Variable globules (size & color), negative network, shiny white lines | Symmetric | Brown, gray, black | Less prominent |
| Reed nevus (active) | Starburst | Central dark pigmentation, peripheral streaks/pseudopods | Symmetric (mandatory) | Dark brown/black/blue | Not prominent |
| Reed nevus (senescent) | Homogeneous or reticular | Structureless pigmentation OR superficial black network + bluish haze | Symmetric | Gray-blue, gray-black, brown | Not prominent |
| Spitz/Reed (early) | Dense globular | Dense globules in early nevogenesis | Symmetric | Variable | Variable |
| Spitz/Reed (multicomponent) | Multicomponent | Combination of starburst + globular + dotted vessel features | May be asymmetric | Variable | Variable |
| Atypical Spitz tumor | Multicomponent (44%) or nonspecific (20%) or typical spitzoid (20%) | Variable: globules, depigmentation, hairpin vessels, shiny white streaks | Variable | Variable | Variable |
| Spitzoid melanoma | Multicomponent/asymmetric | Streaks, globules, dotted vessels, negative network, shiny white lines --- all in disorganized distribution | Usually asymmetric | Multiple colors | Variable |
6. Spitzoid Lesion Management Algorithm (Text-Based Flowchart)
SPITZOID-LOOKING LESION IDENTIFIED
(Features: streaks, dotted vessels, negative network, globules, starburst)
|
v
========================================
STEP 1: DERMOSCOPIC SYMMETRY ASSESSMENT
========================================
|
+------+------+
| |
ASYMMETRIC SYMMETRIC
/DISORGANIZED /ORGANIZED
| |
v v
--> EXCISE ========================
(rule out STEP 2: PATIENT AGE
melanoma) ========================
|
+-----+-----+
| |
AGE >= 12 AGE < 12
| |
v v
--> EXCISE ==============================
or closely STEP 3: CLINICAL MORPHOLOGY
MONITOR ==============================
|
+------+------+
| |
NODULAR FLAT/MACULAR
| |
v v
--> EXCISE or --> MONITOR
CLOSE MONITOR - Every 3 months
- Every 15 days - Prolong intervals
initially if stable
- Excise if - Terminate after
asymmetric 6 months stability
growth - Alternative: ignore
- Prolong to in prepubertal
1 mo, then patients
2 mo if - Alternative: excise
symmetric while small
- Terminate
after 6 mo
stability or
involution
MONITORING TERMINATION CRITERIA:
- Stabilization: No detectable changes between visits >= 6 months apart
- Involution: Complete or partial disappearance
WARNING: ~20% of symmetric Spitz/Reed nevi in children are
ultimately excised due to asymmetric changes during follow-up.
None proved to be AST or melanoma in referenced study.
7. Clinical Pearls
The starburst pattern does not mean Reed nevus exclusively. While classically associated with Reed nevus, the starburst pattern is actually the most common pattern across all Spitz nevi (51% of cases in the IDS systematic review). It represents a specific evolutionary stage, not necessarily a specific histologic subtype.
Negative network + shiny white lines = think Spitz. The co-existence of negative network and shiny white lines is a highly useful diagnostic clue. This combination is NOT seen in classic dermal globular nevi, making it nearly pathognomonic for pigmented Spitz nevi.
Globule variability is the key to distinguishing pigmented Spitz from dermal nevi. Classic dermal globular nevi have monomorphous, uniformly brown globules. Pigmented Spitz nevi have globules that vary in size and color (brown, gray, black).
Symmetry is not a guarantee of benignity. Approximately 13% of dermoscopically symmetric spitzoid lesions in patients over 12 years of age are melanomas. Age must be integrated into every management decision.
Age 12 is the critical threshold. This age serves as the practical dividing line in management. Above 12 years, even symmetric spitzoid lesions carry enough melanoma risk to warrant excision or very close monitoring. Below 12, melanoma is exceedingly rare, permitting more conservative approaches.
Dermoscopic atypia does not equal histologic atypia. A Spitz nevus with a frightening multicomponent pattern on dermoscopy may prove histologically benign. Conversely, a dermoscopically bland-appearing lesion may harbor atypia. Dermoscopy and histopathology are complementary, not interchangeable.
The starburst pattern means active growth. When you see a starburst pattern, the lesion is in its radial growth phase. Senescent Spitz/Reed nevi transition to homogeneous or reticular patterns, at which point they may become difficult to identify as Spitz/Reed nevi.
Multicomponent pattern = biopsy, regardless. Any pigmented lesion displaying a multicomponent pattern should always be biopsied, whether suspected to be a Spitz/Reed nevus or melanoma.
ASTs can perfectly mimic classic Spitz nevi. Twenty percent of ASTs display a nonpigmented typical spitzoid pattern (dotted vessels with or without reticular depigmentation), making them dermoscopically indistinguishable from benign Spitz nevi.
The monitoring dilemma in children is unresolved. Roughly 20% of monitored symmetric Spitz/Reed nevi in children eventually require excision due to asymmetric changes, yet none proved to be malignant in published data. Some experts argue flat, pigmented, symmetric Spitz/Reed nevi in prepubertal patients should simply be ignored rather than subjected to serial monitoring. Others advocate early excision while the lesion is small. There is no consensus.
Clinical Vignettes
Clinical Scenario A 7-year-old girl presents with a 6 mm pink, dome-shaped papule on the left cheek that appeared 2 months ago and has been growing. Dermoscopy reveals multiple regularly distributed dotted vessels surrounded by crisscrossing shiny white lines forming a reticular white network. The pattern is symmetric. No pigment network, globules, or blue-white structures are visible.
What is the most likely diagnosis?
Diagnosis: Classic (nonpigmented) Spitz nevus.
The combination of dotted vessels with shiny white lines forming a reticular white network is characteristic of Spitz nevi (Clinical Pearl 2: negative network + shiny white lines = think Spitz). The symmetric arrangement, young age, and facial location are all typical for Spitz nevus. In a child under 12, the risk of melanoma is exceedingly low (Clinical Pearl 5), supporting conservative management. However, this symmetric pattern can also be seen in 20% of atypical Spitz tumors (Clinical Pearl 9), making them dermoscopically indistinguishable from benign Spitz nevi. Management options include monitoring or excision while the lesion is small -- there is no consensus for prepubertal children (Clinical Pearl 10).
Clinical Scenario A 25-year-old woman presents with a 5 mm heavily pigmented, well-circumscribed papule on the left thigh that appeared 6 weeks ago. Dermoscopy reveals a starburst pattern with symmetrically arranged peripheral streaks (pseudopods and radial lines) radiating from a blue-black homogeneous center. The streaks are distributed uniformly around the entire circumference.
What is the most likely diagnosis?
Diagnosis: Reed nevus (pigmented spindle cell nevus) in the active growth phase -- but excision is recommended given the patient's age.
The starburst pattern with symmetrical peripheral streaks radiating from a blue-black center is the classic presentation of Reed nevus. However, the starburst pattern is not exclusive to Reed nevus -- it is the most common pattern across all Spitz nevi (51% of cases, Clinical Pearl 1). The starburst pattern indicates active radial growth (Clinical Pearl 7). Critically, the patient is 25 years old, well above the age-12 threshold (Clinical Pearl 5). Approximately 13% of dermoscopically symmetric spitzoid lesions in patients over 12 are melanomas (Clinical Pearl 4). Despite the classic pattern, excision is recommended in this age group because symmetry alone does not guarantee benignity.
Clinical Scenario A 16-year-old boy presents with a 9 mm lesion on the left upper arm. Dermoscopy reveals an asymmetric multicomponent pattern with irregular brown-black globules of varying size, focal blue-white veil, an area of negative network, and irregular streaks in the 7 to 10 o'clock sector. The pattern does not show any symmetry axis.
What is the most likely diagnosis?
Diagnosis: Spitzoid lesion requiring excision -- cannot differentiate Spitz nevus from melanoma by dermoscopy.
This case illustrates Clinical Pearl 8: any pigmented lesion displaying a multicomponent pattern should always be biopsied, whether suspected to be a Spitz/Reed nevus or melanoma. The asymmetric distribution of multiple structures (globules of varying size, blue-white veil, negative network, focal streaks) is alarming. While the presence of negative network can be seen in Spitz nevi (Clinical Pearl 2), its combination with asymmetric multicomponent features in a patient over 12 is highly concerning. Additionally, dermoscopic atypia does not always equal histologic atypia, and vice versa (Clinical Pearl 6), so only histopathologic examination can resolve this case. Complete excisional biopsy with adequate margins is mandatory.
9. Cross-References
| Topic | Chapter/Section | Pages (approx.) |
|---|---|---|
| Spitz and Reed nevi --- full chapter | Chapter 7e | 180--186 |
| Starburst pattern --- general description | Chapter 7b (Acquired Melanocytic Nevi) | 157--167 |
| Histopathologic correlates (streaks, pseudopods, globules, negative network) | Chapter 3 | 20--52 |
| Polarized dermoscopy / shiny white lines | Chapters 1--2 | 12--19 |
| Spitzoid melanoma --- extended discussion | Chapter 8e (Other Melanoma Subtypes) | 213--220 |
| Combined nevus (Spitz/Reed + Blue nevus) | Chapter 7d (Blue Nevi) | 175--179 |
| Globular pattern nevi (for comparison) | Chapter 7b | 157--167 |
| Atypical/dysplastic nevi with spitzoid features | Chapter 7b | 157--167 |
10. Related Modules
| Module | Title | Connection |
|---|---|---|
| Module 01 | Introduction and Principles of Dermoscopy | Polarized light required for shiny white lines; equipment basics |
| Module 02 | Histopathologic Correlations | Histopathologic basis of streaks, pseudopods, globules, negative network |
| Module 03 | Pattern Analysis Revised | Fundamental pattern recognition applied to spitzoid lesions |
| Module 04 | Top-Down 2-Step Algorithm | Organized vs. disorganized assessment --- directly applicable to symmetry evaluation in Step 1 |
| Module 05 | Prediction without Pigment | Nonpigmented Spitz nevi share differential with other amelanotic lesions |
| Module 16 | Acquired Melanocytic Nevi | Baseline globular and reticular nevi for comparison; starburst pattern overview |
| Module 18 | Blue Nevi and Variants | Combined Spitz/Reed + blue nevus is described in the blue nevi chapter |
| Module 22 | Nodular Melanoma | Key differential for nodular Spitz nevi in children (Step 3 of management algorithm) |
| Module 25 | Other Melanoma Subtypes (including Spitzoid Melanoma) | Extended discussion of spitzoid melanoma, desmoplastic melanoma, and rare melanoma subtypes |
| Module 26 | Amelanotic and Hypomelanotic Melanoma | Differential for nonpigmented Spitz nevi; polymorphous vessel analysis |
| Module 37 | Digital Monitoring | Sequential digital dermoscopy techniques applicable to monitoring Spitz/Reed nevi in children |
Module 19 --- Version 1.0 Part of the Dermoscopy Educational Course.
Image Sources & Citations
- DERM12345 (Skin Lesion Dataset with 40 Subclasses): DERM12345: A Large-scale Skin Lesion Image Dataset with 40 Subclasses. Figshare. 2023. License: CC-BY-4.0.